Regulated reductive flow through archaeal respiratory systems
Background
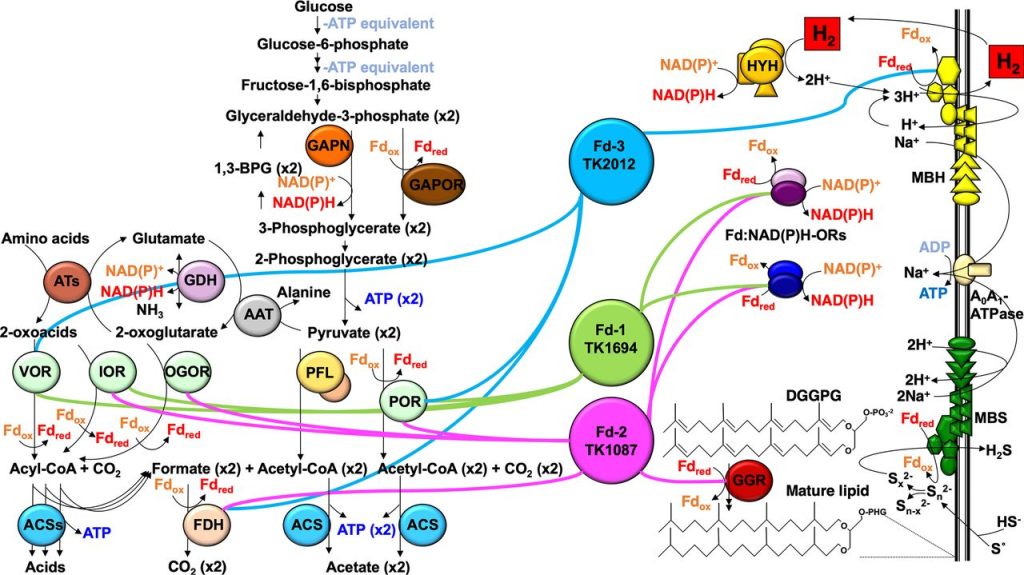
Many Archaea thrive on marginal substrates wherein the catabolic transactions supporting life are barely thermodynamically and energetically profitable. These systems, particularly those present in the methanogens and Thermococcales, are exemplary in that substrates that do not permit even 1 mol of ATP synthesis per mol of substrate can support rapid growth. If reduction of even weakly energetic substrates can be coupled to formation of an electrochemical gradient, this gradient can be exploited for ATP production. ~60-80 kJ/mol are required for ATP production from substrate level phosphorylation in vivo but catabolism of 1 mol of marginal substrates may yield only ~one-tenth of the necessary energy for 1 mol of ATP production. Evolutionary competition has presumably favored mechanisms to conserve and combine multiple, relatively small oG gains to harness sufficient energy to drive ATP synthesis. The Thermococcales, a group of heterotrophic, anaerobic, hyperthermophilic marine archaea use a chemiosmotic mechanism involving an electrochemical ion gradient across the cytoplasmic membrane to drive ATP synthesis via an A1A0 ATP synthase. This gradient is generated via the action of a multi-subunit membrane-bound NiFe-hydrogenase (termed MBH) or a membrane-bound sulfane-reductase (MBS) which couples ferredoxin-driven reduction of protons or polysulfides (thereby generating H2 or H2S, respectively) to H+ translocation across the cytoplasmic membrane. Ferredoxins are critical to this lifestyle, given that their redox potentials (E0’) are extremely low, ~500 mV. This low potential electron carrier is sufficiently energetically rich to not only permit reduction of protons to H2 (E0’ = -414 mv), but to drive such a reaction with sufficient excess that energy is available to drive translocation of ions across the membrane. The resultant chemiosmotic gradient can then be exploited for ATP synthesis.
RELEVANT PUBLICATIONS
- Scott KA, Williams SA, Santangelo TJ. Thermococcus kodakarensis provides a versatile hyperthermophilic archaeal platform for protein expression. Methods in Enzymology. 2021. 659:243-273.
- Burkhart BW, Febvre HP, Santangelo TJ. Distinct physiological roles for the three ferredoxins encoded in the hyperthermophilic archaeon Thermococcus kodakarensis. mBio. 2019. 10(2):e02807-18.
- Gehring AM, Sanders TJ, Santangelo TJ. Markerless Gene Editing in the Hyperthermophilic Archaeon Thermococcus kodakarensis. Bio Protoc. 2017 Nov 20;7(22).
- Santangelo TJ, Cubonova L, Reeve JN. Deletion of alternative pathways for reductant recycling in Thermococcus kodakarensis increases hydrogen production. Mol Microbiol. 2011. Aug;81(4):897-911.
